Technology
Problems to Be Solved
01Challenges in Cell Therapy
In recent years, there has been significant progress in the practical and clinical application of cell therapy. However, products based on adult cells and adult stem cells have been predominant, and constraints such as the limited number of usable cells pose significant obstacles to the broader clinical application for diverse patient populations. In contrast, pluripotent stem cells, including induced pluripotent stem (iPS) cells, offer the potential to differentiate into various cell types and can proliferate in an almost unlimited manner, holding immense promise for the widespread adoption of cell therapy.
On the flip side, ensuring the safety of pluripotent stem cells is crucial. The unlimited proliferative potential of pluripotent stem cells comes with the risk of proliferating like a tumor in the transplanted organism (tumorigenic risk). Even in cases where they do not proliferate, if the transplanted cells fail to exhibit the expected function, there is no mechanism to eliminate the administered cells. This poses a significant challenge in the field of cell therapy.
02Challenges in Gene Therapy
In addition to cell therapy, gene therapy has shown significant advancement in recent years. Ex vivo gene therapy, involving the introduction of genes into cells outside the body, has achieved considerable success, particularly in the treatment of blood cancers. Furthermore, since the introduction of CRISPR/Cas9 genome editing technology in 2012, in vivo gene therapy, directly rewriting genes within the body, has been practically applied. This gene therapy primarily employs viruses as gene carriers and holds great promise as a fundamental treatment for diseases.
However, a crucial aspect of in vivo gene therapy is ensuring the delivery of genes to the required sites. Since viruses lack inherent migratory capabilities, a challenge arises from their insufficient accumulation in diseased areas, such as invasive tumors.

Our Solutions
We are addressing these challenges in cell therapy and gene therapy by utilizing genome-edited induced pluripotent stem (iPS) cells.
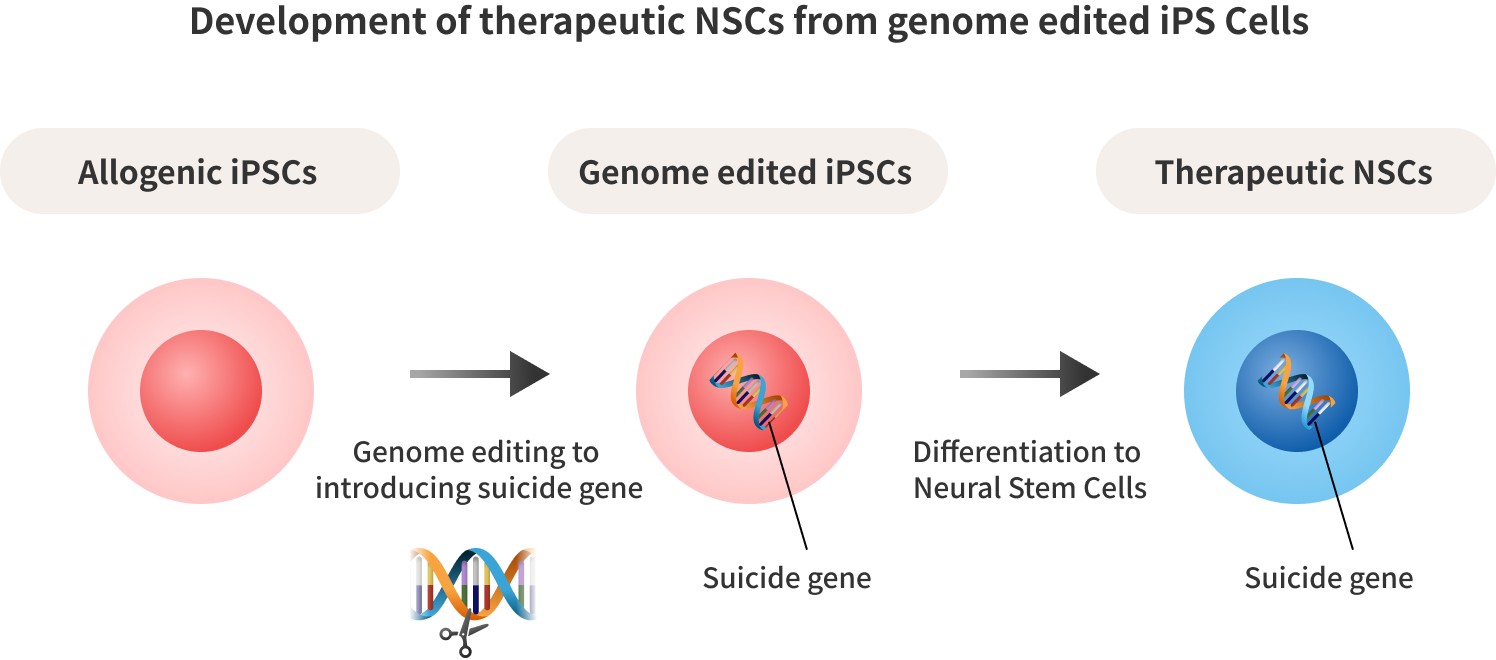
For the treatment of intractable brain diseases, we have developed therapeutic Neural Stem Cells (NSCs) by introducing the CD-UPRT gene into iPS cells through genome editing and inducing their differentiation. In addition to the enhanced productivity achieved with iPS cells, we have introduced a suicide gene that converts prodrugs into cell-killing drugs, ensuring a high level of safety during the administration of cells into the brain.
Moreover, given the inherent ability of neural stem cells to sense and accumulate various chemokines secreted from brain tumors and damaged brain sites, they can serve as effective gene delivery vehicles.
We are advancing the development of therapeutic NSCs, integrating genome editing technology and iPS cell technology, as a novel solution for malignant brain tumors and brain dysfunction.
Our Strength
-
01
High Efficacy
Our therapeutic NSCs have high therapeutic efficacy in brain diseases. Furthermore, they detect chemokines released from tumors and immune cells, migrate toward tumors, and accumulate in brain tumors or brain injury sites. As a result, therapeutic NSCs are expected to show high therapeutic efficacy at the site of the disease.
-
02
High Productivity
Neural stem cells (NSCs) are expected to be an excellent candidate for cell therapy, but their clinical application has been limited due to the difficulty in obtaining human-derived cells and the difficulty in stable production. Our therapeutic NSCs are produced from iPS cells using clinically applicable differentiation technologies, which enables stable quality and mass production for commercial scale.
-
03
High Safety
Although iPS cells have high productivity, the risk of tumorigenesis is a major issue for their clinical application. To avoid this risk, our therapeutic NSCs contain a gene that converts prodrug to tumor-damaging drug (suicide gene). Even if an implanted therapeutic NSC becomes tumorous, it can be safely eliminated from the body by administering the prodrug. In other words, they are iPS cells with a so-called safeguard built into them.
Target Diseases
We are developing treatments for the following two refractory diseases.
